.jpg)
Its structure resembles a single layer of graphite. It can hybridize in sp, sp 2, or sp 3 forms. Video Test 1 2 3 4 Fullerenes Graphene Graphene is another form of the element carbon. A one dimensional fullerene (a convex cage of atoms with only hexagonal and/or pentagonal faces) with a cylindrical shape. The present results could be useful to guide the design of excellent interfaces of mixed-dimensional hybrid carbon materials for various optoelectronic applications. Carbon is the chemical element with atomic number 6 and has six electrons which occupy 1 s 2, 2 s 2, and 2p 2 atomic orbital. Interestingly, electrons either remaining on CNT65 or transferred to C 70 are trapped in the higher conduction band for a while, similarly, due to slow inter-band relaxation. By contrast, in C high E 22 excitation still can lead to ultrafast electron transfer to C 70, but only a comparable amount of electrons are transferred ( ca. Their structures are based on hexagonal rings of carbon atoms joined by covalent bonds. Differently, high E 22 excitation does not induce electron injection to C 60 in C instead, “hot” electrons produced within CNT65 will be trapped in its higher conduction band for a while because of slow inter-band relaxation. Fullerenes are molecules of carbon atoms with hollow shapes. This process is ultrafast and completed within about 200 fs, which is consistent with recent experiments. 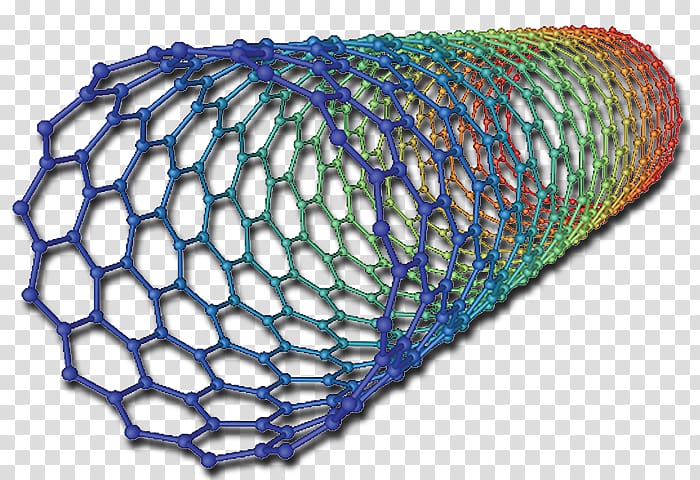
In this contribution, we have employed density functional theory (DFT) and DFT-based nonadiabatic dynamics methods to explore photoinduced interfacial electron transfer processes at interfaces between a single-walled carbon nanotube with chiral index (6,5) and C 60 or C 70 (C and C We have found that with low E 11 excitation, electron transfer takes place from CNT65 to C 60 and C 70 in both heterojunctions. Fullerenes Nanotubes and Carbon Nanostructures The highest Journals Impact IF of Fullerenes Nanotubes and Carbon Nanostructures is 2.06. Initial atomic coordinates (u, v, w), chiral vector (n, m) and calculated atomic coordinates (u’, v’, w’) are obtained from the output files.Hybrid carbon materials are found to exhibit novel optoelectronic properties at their interfaces, but the related interfacial carrier dynamics is rarely explored theoretically. 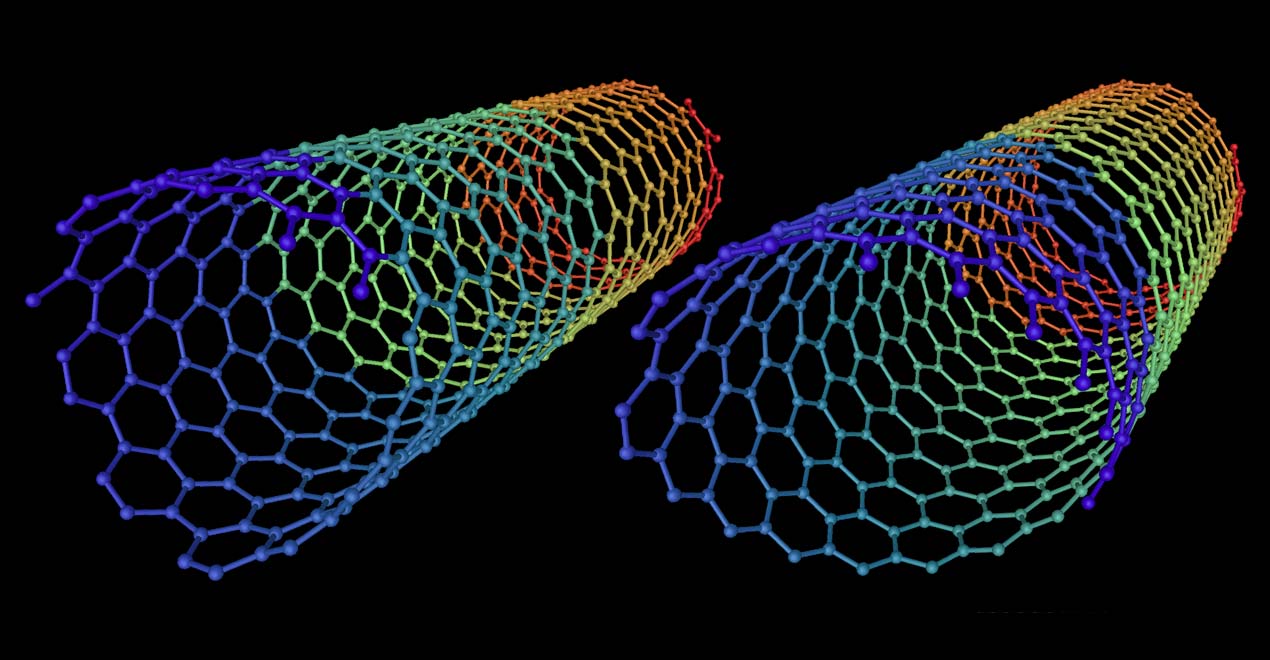
Nanotubes are large, linear fullerenes with aspect. 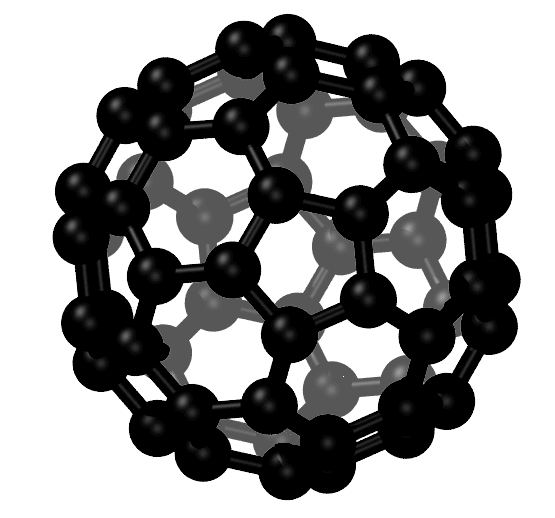
To finalize the computation, CASTEP uses a parameter named as elec_energy_tol (electrical energy tolerance) (default 1x10-5 eV) which represents that the change in the total energy from one iteration to the next remains below some tolerance value per atom for a few self-consistent field steps. Fullerenes are close-caged molecules contain- ing only hexagonal and pentagonal interatomic bonding networks. CNT calculation parameters are used as default parameters. The atom type is selected as carbon, bond length is used as 1.42 A° (default value). Twenty-five years on from the discovery of C 60, the outstanding properties and potential applications of the synthetic carbon allotropes fullerenes, nanotubes and graphene overwhelmingly. A comprehensive offering of carbon nanomaterials from fullerene, to carbon nanotubes, graphene, carbon quantum dots and nanodiamonds to empower your research and development in energy storage, high performing electronics, smart composites, and novel nano therapeutics. Regardless of the noticeable difference in the fabrication of CNTs, fullerenes and graphene, their physical and chemical properties are related to each other. Different chiral vectors are used for each CNT simulation. Another new member of the carbon family is the carbon nanotubes (CNTs). Initial coordinates of all carbon atoms are generated randomly. Many CNTs are simulated in CASTEP, then geometry optimizations are calculated. The dataset is generated with CASTEP using CNT geometry optimization. Despite the impressive intrinsic properties, challenges in processing and chemical modification continue to hinder applications. Comparison of small covalent molecules and giant covalent molecules. Since the discovery of buckminsterfullerene over 30 years ago, sp2-hybridised carbon nanomaterials (including fullerenes, carbon nanotubes, and graphene) have stimulated new science and technology across a huge range of fields. DFT is the most successful method calculates atomic coordinates faster than other mathematical approaches, and it also reaches more accurate results. Carbon nanotubes are used to reinforce materials in tennis racquets. CASTEP can simulate a wide range of properties of materials proprieties using density functional theory (DFT).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
